 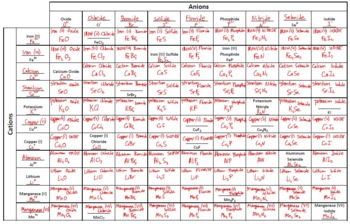 
Polyatomic anions typically have common names that you must learn some examples are in Table 2.4. Because the charges on these ions can be predicted from their position in the periodic table, it is not necessary to specify the charge in the name. If you have a polyatomic ion, use the Common Ion table to find. Write the symbol and charge for the non-metal. If you look at a salt crystal you will see that each positive ion is surrounded on all 6 sides by negative ions, and that a crystal lattice forms. The charge is the Roman Numeral in parentheses. 
The anion is named by taking the elemental name, removing the ending, and adding -ide. For example, K+1 K + 1 is called the potassium ion, just as K K is called the potassium atom. The cation has the same name as its element. Therefore, a sample of calcium chloride contains twice as many chloride ions as calcium ions. An ionic compound is named first by its cation and then by its anion. Monatomic anions are named by adding the suffix - ide to the root of the name of the parent element thus, Cl − is chloride, O 2− is oxide, P 3− is phosphide, N 3− is nitride (also called azide), and C 4− is carbide. When writing the formula of an ionic compound we use the lowest whole number ratio of cations ( + ions) to anions ( - ions). Calcium Chloride is an ionic compound with the chemical formula CaCl2. The names of the common polyatomic cations that are relatively important in ionic compounds (such as, the ammonium ion) are in Table 2.4. †The isolated mercury(I) ion exists only as the gaseous ion. Identify the ions magnesium hydroxide contains magnesium. Trick: Set of Anions Charge of Cation and set of Cations Charge of Anion Beware that this must be the lowest whole ratio of cation to anion. Sometimes an ionic formula contains compound ions a group of atoms bonded together with an overall charge. \) Common Cations of Metals That Form More Than One Ion Cation Ionic Compound Formula and the Principle of Charge Neutrality: The formula of an ionic compound represents the lowest whole number ratio of cations to anions, it is as simple as that. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
